Ampoule Filling Lines
In the pharmaceutical industry, Ampoule Filling & Sealing Machines are used to process ampoules, which are small, airtight glass containers designed to maintain the sterility and integrity of their contents. Commonly used for storing injectable medications, vaccines, and sensitive liquids or gases, ampoules protect products from air, moisture, and contaminants. They are sealed to ensure product stability until the moment of use.
Ampoules come in two main types: open ampoules, which are ready for filling and sealing, and closed ampoules, which require an opening step before filling. The use of Ampoule Filling & Sealing Machines is critical in ensuring safe, precise, and contamination-free drug delivery in pharmaceutical manufacturing.
Filling Line Comes Under a Different Contentment System

ORABS – Open Restricted Access Barrier System
ORABS provides a physical barrier typically constructed from tempered glass or acrylic panels that separates the critical product zone from operators, while still allowing controlled manual intervention when necessary
Unidirectional Laminar Airflow (LAF): HEPA-filtered airflow maintains Grade A conditions over the filling area.
Glove Ports: Enable operator access without direct exposure, reducing contamination risk.
Grade B Cleanroom Installation: Operates within a Grade B background environment as per EU GMP standards.
Controlled Manual Interventions: Permits aseptic manual access with defined risk control procedures.
- Ampoule Rotary Washing Machine
- Sterilisation & Depyrogenation Tunnel
- Filling Line
- Sealing Mechanis
- Labelling Machine
Ampoule Rotary Washing Machine
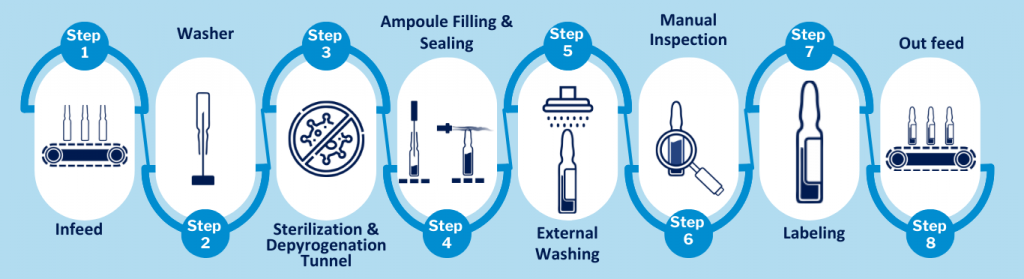
The Ampoule Rotary Washing Machine is installed at the beginning of the filling line and is designed to thoroughly clean the Ampoule by removing particles and contaminants before filling and sealing operations.
The machine carries out a 6-stage washing and rinsing process to achieve pharmaceutical-grade cleanliness:
- Recirculated Water & Air: Ampoules are initially washed with recirculated water and then blown with compressed air to remove loose particles.
- Purified Water & Air: A second washing stage uses purified water followed by sterile compressed air for improved cleaning.
- Water for Injection (WFI): The final rinsing stage is performed with WFI to ensure the highest level of sterility.
- Sterile Air Drying: After washing, ampoules are completely dried using sterile compressed air.
Optional Features (as per process requirements):
- Ultrasonic Washing: An optional ultrasonic unit can be integrated for applications requiring enhanced cleaning performance. High-frequency sound waves remove microscopic contaminants from the Ampoule surface.
- Siliconization: When required, a siliconization system can be added to apply a fine mist of pharmaceutical-grade silicone inside the Ampoule to improve internal surface smoothness.
Sterilisation & Depyrogenation Tunnel
After the Ampoules are washed, they are transferred into the Sterilisation & Depyrogenation Tunnel, a critical unit in the aseptic processing line that ensures Ampoule are sterile and free from pyrogens (endotoxins).
The tunnel is divided into three functional zones, each designed for a specific process step:
- Drying Zone (Pre-heating Zone):
In this zone, residual moisture on the inner and outer surfaces of the Ampoule is completely evaporated. Proper drying prevents any interference with sterilisation and avoids dilution or contamination during the subsequent filling process. - Hot Zone (Sterilisation & Depyrogenation Zone):
This is the core section of the tunnel and operates at high temperatures, typically above 300°C. Laminar airflow combined with HEPA filtration maintains ISO 5 (Class 100) conditions, ensuring sterility throughout the process. Ampoule remains in this zone for a validated exposure time to achieve effective sterilisation and depyrogenation. - Cooling Zone:
After sterilisation, Ampoule pass into the cooling zone where filtered, cool air gradually reduces their temperature to ambient conditions. This controlled cooling prevents thermal shock and prepares the Ampoule for direct transfer into the filling area under aseptic conditions.
Ampoule Filling Line
After washing, sterilisation, and depyrogenation, the ampoules are transferred to the filling and sealing section, where the product is filled under strictly controlled aseptic conditions.
Transfer to the Filling Machine:
Clean ampoules are conveyed through a smooth, automated conveyor system to the filling and sealing machine. This ensures gentle handling, prevents damage, and maintains a continuous and efficient production flow without risk of contamination.
Filling System and Product Handling:
The filling operation is carried out using high-precision filling needles designed to deliver accurate and consistent volumes into each ampoule. These needles ensure smooth liquid flow while minimising splashing, foaming, and air entrapment during the filling process.
A product recirculation buffer tank is integrated into the system to maintain uniform product temperature and concentration throughout the filling cycle. Continuous recirculation prevents settling or stratification of the product and ensures consistent dosing across the entire batch.
High-Accuracy Liquid Filling:
The machine uses precision dosing systems such as peristaltic or rotary piston pumps to achieve accurate and uniform filling volumes. This prevents overfilling, underfilling, and air bubble formation that could affect product quality and dosage consistency.
Optional Nitrogen Flushing (Pre & Post Filling):
Nitrogen flushing can be provided as an optional feature based on product requirements. Nitrogen gas may be injected before filling to remove residual air inside the ampoule and after filling to create an inert atmosphere. This option is especially beneficial for oxygen-sensitive formulations, helping to improve product stability and extend shelf life.
Ampoule Sealing Machine
After filling, the ampoules move to the sealing station, where they are sealed to protect the product and maintain sterility.
Flame-Based Glass Sealing:
At the sealing station, the ampoule neck is exposed to a precisely controlled flame that softens and melts the glass, forming a tight and uniform seal. This creates an airtight closure that prevents contamination and leakage.
Preheating and Burner Control:
Before final sealing, the ampoule tip is gently preheated to reduce thermal stress on the glass and ensure a smooth, consistent sealing process. Burners are accurately positioned to focus heat only on the ampoule neck without affecting other areas.
Oxygen and LPG Flame System:
The sealing flame is generated using a controlled mixture of oxygen and LPG (liquefied petroleum gas). This produces a clean, high-temperature flame ideal for glass melting. Flame intensity and direction are precisely regulated to achieve consistent sealing quality across all ampoules.
Ampoule Rotary Labelling Machine
Ampoules are fed into a rotary labelling system where each Ampoule is securely held and rotated for precise, wrinkle-free label application, ensuring proper alignment and scannability.
The machine automatically applies pre-printed self-adhesive labels at high speed, delivering high-throughput and consistent accuracy across batches.
Each label carries critical information, including:
- Product name
- Dosage
- Batch number
- Manufacturing and expiry dates
- Barcodes or 2D matrix codes for serialization and traceability
Product Flow